One of the early models of atoms, called the Thomson atomic model, has been compared to these food items in terms of appearance. Its influence echoed throughout other scientific and cultural spheres, engraving its place in the annals of atomic theory’s history.You all must have seen a watermelon, a raisin pudding and a plum pudding in your daily lives. 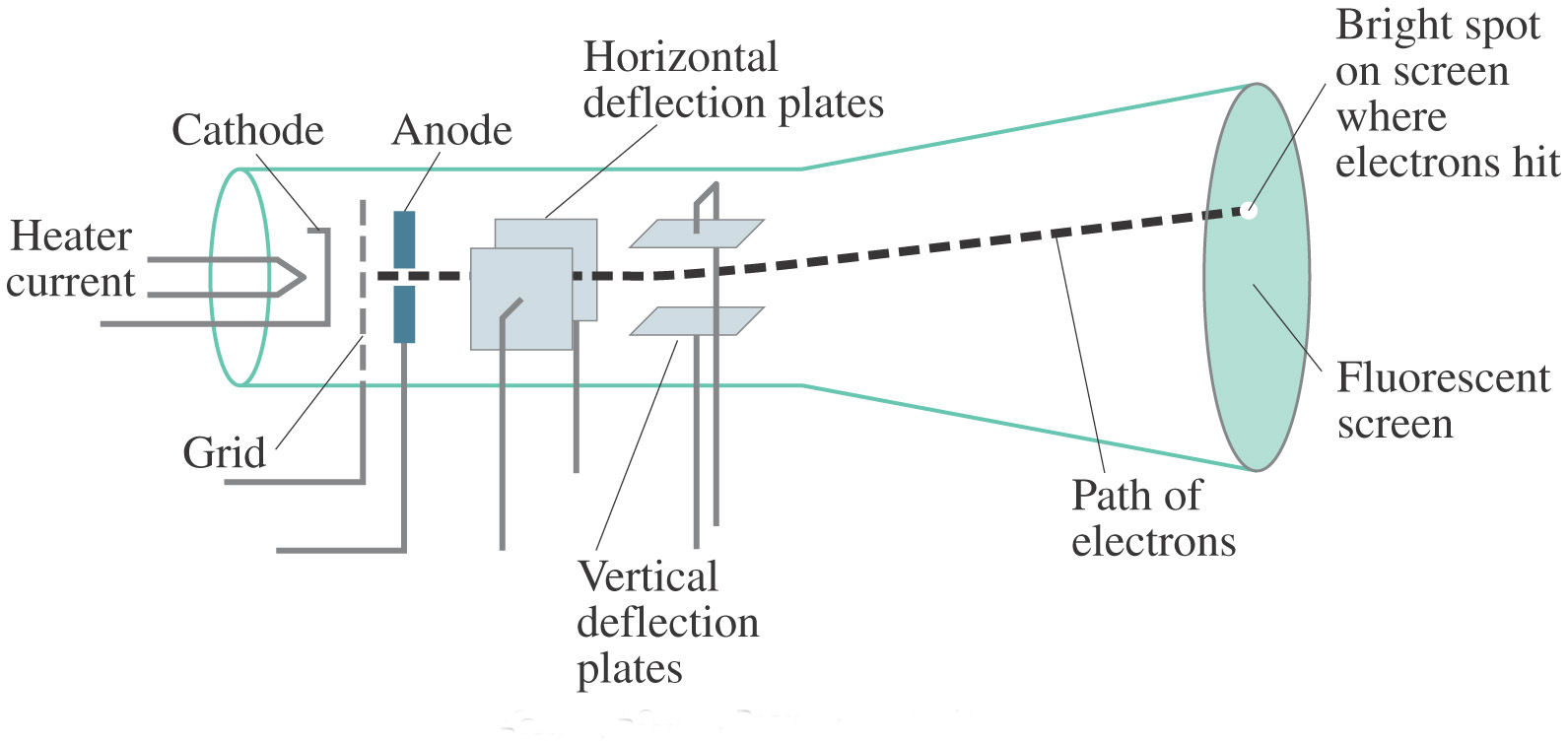
It acted as a source of inspiration for Niels Bohr, who developed his quantum model of the atom, successfully integrating classical and quantum mechanics.ĭespite its eventual replacement by more accurate models, Thomson Atomic Model retains historical and scientific significance. Instead, it revealed the existence of internal structures and subatomic particles within atoms, steering new areas of exploration into the nature of matter and energy.įurthermore, the model’s impact was not confined to physics alone. It challenged the idea that atoms are indivisible and unchangeable entities, a belief held by ancient philosophers. Moreover, the model’s significance extended beyond its limitations. Based on scientific evidence and logical reasoning, it transformed subsequent research and experimentation. Though the Thomson Atomic Model may have proven inaccurate, it was significant in advancing atomic theory and modern physics. It could not explain the presence of isotopes.It could not explain atomic spectra, which are the emission and absorption of specific wavelengths of light by atoms.Thomson’s model did not state the presence of a nucleus in the atom. Rutherford’s experiment revealed that most of an atom’s mass and positive charge is concentrated in a minute, dense nucleus at the center of the atom. It could not account for the outcomes of Rutherford’s gold foil experiment.In other words, it could not justify the atom’s stability. It did not explain how the positive charge contained the negatively charged electrons.Instead, the model presumed a uniform positive charge distribution throughout the atom. It failed to consider the existence of positively charged particles within the atom, a discovery attributed later to Ernest Rutherford, who identified them as protons.Some of the limitations of Thomson’s Atomic Model are: Thomson’s atomic model had numerous limitations that were ultimately overcome by further experiments and research by various scientists. When the electrons approach the outer part of the atom, the positive charge at the center exceeds the nearby negative charges, causing the electron to be attracted back toward the central region of the atom. 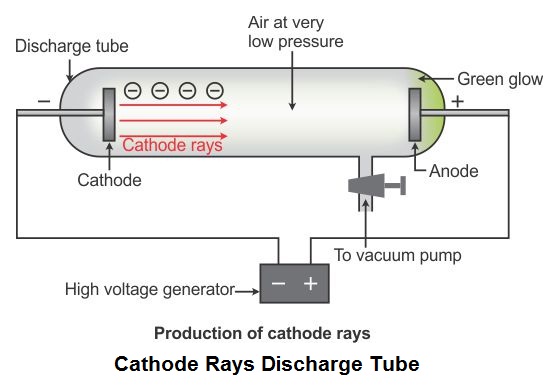
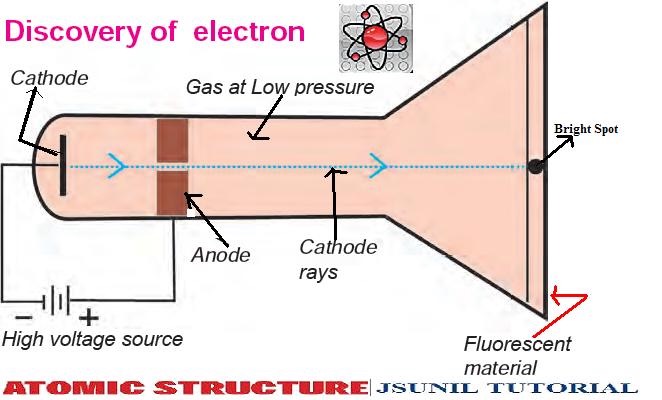
The positive substance was likened to jelly or a thick soup, and the electrons were believed to possess some degree of mobility. It envisioned electrons being enclosed within a uniform sphere of positive charge, similar to blueberries inserted into a muffin. Thomson’s model became known as the “plum pudding model” or “chocolate chip cookie model”. Nevertheless, Thomson’s model is significant because it introduced the idea that an atom consists of charged particles. Later models established that the atom consists of a nucleus having positively-charged protons and neutral neutrons, with negatively-charged electrons surrounding the nucleus. This model became known as Thomson Atomic Model. He proposed that the atom was a sphere with charges embedded within it. According to his hypothesis, an atom consists of positive and negative charges present in equal amounts so that it is electrically neutral. Thomson proposed a model of the atom in 1904 after doing a series of cathode tube experiments. 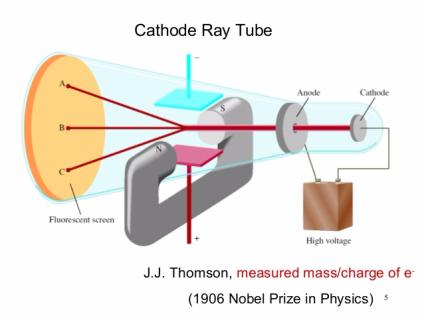
Following his discovery of the electron in 1897, British Physicist J.J.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
